Online Database of Chemicals from Around the World
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Wuhan Comings Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | comings.en.made-in-china.com | |||
 | +86 13659820406 | |||
 | info@comingschemi.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | Bis(4-amino-3-carboxyphenyl)methane |
| Synonyms | 3,3'-Dicarboxy-4,4'-diaminodiphenylmethane; 4,4'-Diamino-3,3'-dicarboxydiphenylmethane; 4,4'-Diaminodiphenylmethane-3,3'-dicarboxylic acid; 5,5'-Methylenebis[2-aminobenzoic acid]; 5,5'-Methylenedianthranilic acid; CB 37136; MBAA; NCI 37136; NSC 37136 |
| Molecular Structure | 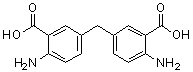 |
| Molecular Formula | C15H14N2O4 |
| Molecular Weight | 286.28 |
| CAS Registry Number | 7330-46-3 |
| EC Number | 230-830-0 |
| SMILES | C1=CC(=C(C=C1CC2=CC(=C(C=C2)N)C(=O)O)C(=O)O)N |
| Solubility | Practically insoluble (0.074 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.442±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 262 °C (water acetic acid ) (Expl.) |
| Boiling point | 576.4$+/-$50.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 302.4$+/-$30.1 $degree$C (Calc.)* |
| Index of refraction | 1.714 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
|
Bis(4-amino-3-carboxyphenyl)methane is an aromatic diamine compound in which two benzene rings are connected via a central methylene (–CH2–) group, with each ring substituted by a primary amino group at the 4-position and a carboxyl group at the 3-position. This structure combines nucleophilic amino groups with acidic carboxyl functionalities, making it useful as an intermediate in polymer synthesis, organic reactions, and the preparation of functionalized aromatic derivatives. The combination of electron-donating amino groups and electron-withdrawing carboxyl groups allows for controlled reactivity and facilitates condensation and crosslinking reactions. The molecular structure consists of a central methylene bridge linking two para-amino- and meta-carboxyl-substituted benzene rings. The amino groups act as nucleophilic centers capable of participating in acylation, sulfonylation, diazotization, and polycondensation reactions. The carboxyl groups provide sites for esterification, amide formation, or salt formation, which can influence solubility, polymerization behavior, and thermal stability. The methylene bridge introduces flexibility between the aromatic rings, which can affect the physical properties of materials derived from this compound. Synthesis of bis(4-amino-3-carboxyphenyl)methane typically involves condensation of 4-nitro-3-carboxybenzaldehyde derivatives with formaldehyde, followed by reduction of nitro groups to yield the diamine. Reaction conditions are carefully controlled to achieve high regioselectivity, avoid over-condensation, and ensure complete reduction of nitro groups without affecting carboxyl functionalities. The resulting compound is generally obtained as a solid with good stability and purity, suitable for further chemical transformations. One of the main applications of this compound is as a monomer or intermediate in polymer synthesis. Its amino and carboxyl functionalities allow it to form polyamides, polyimides, and other high-performance polymers. The para-amino groups enable chain extension and crosslinking, while the carboxyl groups can be converted to esters or amides to modify solubility, thermal behavior, or mechanical properties. Polymers derived from this diamine are used in adhesives, coatings, electronic materials, and composites where chemical resistance and mechanical strength are required. In addition to polymer applications, bis(4-amino-3-carboxyphenyl)methane is used in organic synthesis for the preparation of functionalized aromatic compounds, dyes, and heterocycles. Its bifunctional nature enables selective chemical reactions at amino or carboxyl positions, allowing for the design of molecules with targeted properties for chemical, material, or pharmaceutical research. Overall, bis(4-amino-3-carboxyphenyl)methane is a versatile aromatic diamine with broad utility in polymer chemistry, organic synthesis, and functional material development. Its combination of para-amino and meta-carboxyl substituents, together with the methylene bridge, provides both chemical reactivity and structural flexibility, making it an important intermediate for high-performance materials and advanced chemical compounds. References 2023. Self-healing hybrid hydrogels with sustained bioactive components release for guided bone regeneration. Journal of Nanobiotechnology. DOI: 10.1186/s12951-023-01811-8 2022. A quantitative high-throughput screen identifies compounds that lower expression of the SCA2-and ALS-associated gene ATXN2. The Journal of biological chemistry. DOI: 10.1016/j.jbc.2022.102228 2022. Discovery of small molecule inhibitors of Plasmodium falciparum apicoplast DNA polymerase. Journal of Enzyme Inhibition and Medicinal Chemistry. DOI: 10.1080/14756366.2022.2070909 |
| Market Analysis Reports |