Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Mascot I.E. CO.,Ltd. | China | |||
|---|---|---|---|---|
 | www.mascotchem.com | |||
 | +86 (519) 8501-0339 +86 13584504415 | |||
 | +86 (519) 8501-0366 | |||
 | info@mascotchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2006 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Changzhou Xiangren Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemxr.com | |||
 | +86 (519) 8285-0370 | |||
 | lxsroad@yahoo.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Yishenghui Bio-medical Technologies Inc. | China | |||
|---|---|---|---|---|
 | www.ychemical.com | |||
 | +86 (21) 6145-3981 +86 13564731332 | |||
 | +86 (21) 6145-3981 | |||
 | yishenghui99@126.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Jintan Xinwang Chhemical Technolgy Service Center | China | |||
|---|---|---|---|---|
 | www.jtxwchem.com | |||
 | +86 (519) 8280-8245 8261-2034 +86 13921020245 | |||
 | +86 (519) 8280-8245 / 8261-0403 | |||
 | sale@jtxwchem.com jtxwchem@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| Jintan Sida Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.star-chem.com | |||
 | +86 (519) 8233-7678 +86 13906146788 | |||
 | +86 (519) 8233-6758 | |||
 | whp@star-chem.com | |||
| Chemical manufacturer | ||||
| Marshallton Research Laboratories | USA | |||
|---|---|---|---|---|
 | www.marshalltonlabs.com | |||
 | +1 (336) 983-2131 | |||
 | +1 (336) 983-0096 | |||
 | marshallton@windstream.net | |||
| Chemical manufacturer since 1974 | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic ketone |
|---|---|
| Name | 2'-Fluoroacetophenone |
| Synonyms | 1-(2-Fluorophenyl)ethanone |
| Molecular Structure | 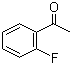 |
| Molecular Formula | C8H7FO |
| Molecular Weight | 138.14 |
| CAS Registry Number | 445-27-2 |
| EC Number | 207-156-0 |
| SMILES | CC(=O)C1=CC=CC=C1F |
| Density | 1.1$+/-$0.1 g/cm3 Calc.*, 1.137 g/mL (Expl.) |
|---|---|
| Boiling point | 180.8 $degree$C 760 mmHg (Calc.)*, 187 - 189 $degree$C (Expl.) |
| Flash point | 61.7 $degree$C (Calc.)*, 60 $degree$C (Expl.) |
| Index of refraction | 1.491 (Calc.)*, 1.507 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P280-P305+P351+P338 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 2810 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
2′-Fluoroacetophenone is an aromatic ketone belonging to the class of fluorinated acetophenone derivatives. Its molecular formula is C8H7FO, and its structure consists of an acetophenone framework in which a fluorine atom is substituted at the 2-position (ortho position) of the benzene ring relative to the acetyl group. Structurally, the molecule contains two principal functional components: an aromatic benzene ring and a methyl ketone group (–COCH3). The fluorine substituent is attached directly to the aromatic ring adjacent to the carbonyl-containing acetyl substituent. This arrangement creates a conjugated aromatic ketone system whose electronic properties are strongly influenced by the fluorine atom. The carbonyl group is the primary reactive site of the molecule. As a ketone, it can undergo nucleophilic addition reactions, condensation reactions such as aldol condensations, reduction to secondary alcohols, and formation of oximes, hydrazones, and imines. The conjugation between the aromatic ring and the carbonyl group stabilizes the structure and affects spectroscopic behavior. Fluorine substitution significantly alters the electronic characteristics of the aromatic ring. Fluorine is highly electronegative and exerts a strong inductive electron-withdrawing effect, although it can also donate electron density weakly through resonance interactions. In the ortho position, the fluorine atom influences both the reactivity and steric environment around the carbonyl group. The ortho arrangement of the fluorine substituent relative to the acetyl group introduces steric and electronic interactions that can affect molecular conformation and reaction selectivity. The carbonyl oxygen and fluorine atom may participate in weak intramolecular interactions that influence the preferred geometry of the molecule. Fluorinated aromatic ketones such as 2′-fluoroacetophenone are important intermediates in synthetic organic chemistry. The fluorine atom can influence biological activity, metabolic stability, lipophilicity, and binding interactions in pharmaceutical compounds. Consequently, fluorinated acetophenones are widely used as precursors in medicinal chemistry and agrochemical synthesis. The compound can serve as a starting material for synthesis of heterocycles, fluorinated alcohols, amines, and substituted aromatic derivatives. The ketone functionality is highly versatile and enables numerous carbon–carbon and carbon–heteroatom bond-forming reactions. Common synthetic routes to 2′-fluoroacetophenone include Friedel–Crafts acylation of fluorobenzene with acetylating agents such as acetyl chloride or acetic anhydride in the presence of Lewis acid catalysts. Alternatively, selective fluorination of acetophenone derivatives may also be employed. From a physicochemical perspective, 2′-fluoroacetophenone is moderately polar because of the carbonyl group, while the aromatic ring contributes hydrophobic character. The fluorine atom increases dipole moment and can influence intermolecular interactions without substantially increasing molecular size. The aromatic ketone chromophore gives rise to characteristic ultraviolet absorption due to π–π* and n–π* electronic transitions. The fluorine substituent can subtly shift these absorption properties relative to unsubstituted acetophenone. Historically, fluorinated aromatic compounds became increasingly important in the second half of the 20th century as organofluorine chemistry expanded in pharmaceutical, agrochemical, and materials research. Introduction of fluorine into aromatic systems often provides improved chemical and biological properties compared with nonfluorinated analogues. Overall, 2′-fluoroacetophenone is an ortho-fluorinated aromatic methyl ketone characterized by a conjugated acetophenone core and a fluorine substituent adjacent to the carbonyl-bearing side chain. Its electronic properties and versatile ketone functionality make it an important intermediate in synthetic and medicinal chemistry. References 2025. Fluorination of Arenes with Reagents Containing an I(III)—F Bond. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-246-00105 2019. An Alkali-tolerant Carbonyl Reductase from Bacillus subtilis by Gene Mining: Identification and Application. Catalysis Letters. DOI: 10.1007/s10562-019-02873-w 2022. Nickel-Catalyzed Dehydrogenative Coupling of 2-Aminobenzyl Alcohol with Aryl Methyl Ketones or Benzyl Cyanides. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-115-00982 |
| Market Analysis Reports |