Online Database of Chemicals from Around the World
| Junkai (Tianjin) Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.junkaichem.com | |||
 | +86 (22) 8568-9515 ex 8818 +86 13920864700 | |||
 | +86 (22) 2378-7875 | |||
 | sales@junkaichem.com jane-jkchem@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2007 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Comings Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | comings.en.made-in-china.com | |||
 | +86 13659820406 | |||
 | info@comingschemi.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Aronis | Russia | |||
|---|---|---|---|---|
 | www.aronis.ru | |||
 | +7 (926) 578-0336 | |||
 | +7 (495) 426-5336 | |||
 | rakishev@aronis.ru | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Alkane |
|---|---|
| Name | 1,1-Bis(4-aminophenyl)cyclohexane |
| Synonyms | 4,4'-cyclohexane-1,1-diyldianiline; {4-[1-(4-aminophenyl)cyclohexyl]phenyl}amine |
| Molecular Structure | 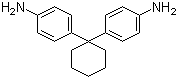 |
| Molecular Formula | C18H22N2 |
| Molecular Weight | 266.38 |
| CAS Registry Number | 3282-99-3 |
| EC Number | 221-926-3 |
| SMILES | C1CCC(CC1)(C2=CC=C(C=C2)N)C3=CC=C(C=C3)N |
| Density | 1.1$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 110 $degree$C (Expl.) |
| Boiling point | 450.9$+/-$38.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 271.7$+/-$26.3 $degree$C (Calc.)* |
| Index of refraction | 1.633 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||||||
| Safety Statements | P264-P280-P302+P352+P332+P313+P362+P364-P305+P351+P338+P337+P313 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
1,1-Bis(4-aminophenyl)cyclohexane is an aromatic diamine compound in which two para-substituted amino groups are attached to phenyl rings, and both phenyl rings are connected to the same carbon of a cyclohexane moiety. This structure combines the reactivity of aromatic amino groups with the rigidity of the cyclohexane core, making the compound a key intermediate in polymer synthesis, organic synthesis, and materials chemistry. Its bifunctional nature allows it to act as a monomer for the production of high-performance materials. The molecular structure features a central cyclohexane ring with a quaternary carbon at the 1-position bonded to two 4-aminophenyl groups. The para-oriented amino groups on the phenyl rings provide reactive sites for nucleophilic substitution, acylation, and condensation reactions. The cyclohexane bridge introduces steric bulk and rigidity, which influences both the physical properties of derived materials and the selectivity of chemical reactions involving the amino groups. Synthesis of 1,1-bis(4-aminophenyl)cyclohexane generally involves the hydrogenation of the corresponding bis-nitrophenyl cyclohexane precursor. The nitro groups are reduced to amino groups under controlled conditions to yield the diamine with high purity. The cyclohexane ring is often introduced via condensation of phenol derivatives with cyclohexanone, followed by nitration and reduction. These steps ensure both the correct substitution pattern and preservation of the cyclohexane backbone. One of the primary applications of this compound is in the preparation of polyamides, polyimides, and epoxy resins. Its diamine functionality allows for polymer chain extension and crosslinking, while the rigid cyclohexane core contributes to enhanced thermal stability, mechanical strength, and chemical resistance in the resulting polymers. Such materials are widely used in coatings, adhesives, electronic components, and advanced composites. In addition to polymer applications, 1,1-bis(4-aminophenyl)cyclohexane is employed as an intermediate in organic synthesis. Its amino groups can participate in acylation, sulfonylation, and Schiff base formation, making it a versatile building block for the synthesis of functional aromatic compounds, ligands, and bioactive molecules. The steric influence of the cyclohexane moiety can be exploited to control the geometry of products and influence their reactivity. Overall, 1,1-bis(4-aminophenyl)cyclohexane is a multifunctional aromatic diamine with important applications in polymer chemistry, materials science, and organic synthesis. Its combination of para-oriented amino groups, rigid cyclohexane core, and structural versatility makes it a valuable intermediate for the design and production of high-performance polymers and functional chemical compounds. References 2025. Spontaneous orientation polarization driven by designing molecular asymmetry. Communications Materials. DOI: 10.1038/s43246-025-00815-1 2022. A quantitative high-throughput screen identifies compounds that lower expression of the SCA2-and ALS-associated gene ATXN2. The Journal of biological chemistry. DOI: 10.1016/j.jbc.2022.102228 2022. Discovery of small molecule inhibitors of Plasmodium falciparum apicoplast DNA polymerase. Journal of Enzyme Inhibition and Medicinal Chemistry. DOI: 10.1080/14756366.2022.2070909 |
| Market Analysis Reports |