Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Wuhan Comings Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | comings.en.made-in-china.com | |||
 | +86 13659820406 | |||
 | info@comingschemi.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| AcceleDev Chemical LLC | USA | |||
|---|---|---|---|---|
 | www.acceledev.com | |||
 | +1 (973) 439-1845 | |||
 | +1 (973) 439-1399 | |||
 | info@acceledev.com | |||
| Chemical manufacturer since 2003 | ||||
| Classification | Organic raw materials >> Amino compound >> Cycloalkylamines, aromatic monoamines, aromatic polyamines and derivatives and salts |
|---|---|
| Name | 2,4,6-Trimethyl-1,3-phenylenediamine |
| Synonyms | 2,4,6-Trimethyl-m-phenylenediamine; 2,4-Diamino-1,3,5-trimethylbenzene |
| Molecular Structure | 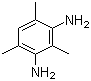 |
| Molecular Formula | C9H14N2 |
| Molecular Weight | 150.22 |
| CAS Registry Number | 3102-70-3 |
| EC Number | 221-456-9 |
| SMILES | CC1=CC(=C(C(=C1N)C)N)C |
| Density | 1.1$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 89 - 91 $degree$C (Expl.) |
| Boiling point | 297.7$+/-$35.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 158.4$+/-$25.4 $degree$C (Calc.)* |
| Index of refraction | 1.605 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS07;GHS08;GHS09 Warning Details GHS07;GHS08;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335-H341-H411 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P318-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
2,4,6-Trimethyl-1,3-phenylenediamine is an aromatic diamine compound in which two amino groups are positioned at the 1- and 3-positions of a benzene ring, while three methyl groups occupy the 2-, 4-, and 6-positions. This substitution pattern provides both steric hindrance and electronic modulation of the amino groups, making the compound a valuable intermediate in dye chemistry, polymer synthesis, and organic synthesis. The combination of nucleophilic amino groups and electron-donating methyl groups enhances its chemical reactivity and solubility characteristics. The molecular structure consists of a benzene ring substituted symmetrically with two amino groups and three methyl groups. The amino groups are nucleophilic and can participate in reactions such as acylation, sulfonylation, and diazotization, while the methyl groups stabilize the aromatic ring and influence the electron density on the amino groups. The steric effects of the methyl groups can also affect the selectivity of reactions, which is important when this compound is used as a building block for complex molecules. Synthesis of 2,4,6-trimethyl-1,3-phenylenediamine typically begins with a methylated nitrobenzene derivative, followed by selective reduction of the nitro groups to form the diamine. Reaction conditions are carefully controlled to maintain the desired substitution pattern and prevent over-reduction or side reactions. The resulting compound is obtained in high purity and is suitable for further chemical transformations. One of the primary applications of this diamine is as an intermediate in the production of azo dyes and pigments. The amino groups can undergo diazotization and subsequent coupling reactions with phenols or other aromatic compounds to form colorful azo dyes with specific spectral properties. The methyl groups enhance solubility and can influence the hue and stability of the dyes. In polymer chemistry, 2,4,6-trimethyl-1,3-phenylenediamine serves as a monomer for high-performance polyamides, polyimides, and epoxy curing agents. The diamine functionality allows for crosslinking or chain extension, while the methyl substituents contribute to the rigidity, thermal stability, and solubility of the resulting polymers. Such materials are used in coatings, adhesives, and composites requiring chemical resistance and mechanical strength. The compound is also used in organic synthesis as a versatile intermediate for preparing heterocyclic compounds, ligands, and functionalized aromatic molecules. Its combination of reactive amino groups and electron-donating methyl substituents makes it a useful reagent for constructing complex molecules in medicinal chemistry and materials science. Overall, 2,4,6-trimethyl-1,3-phenylenediamine is a multifunctional aromatic diamine with significant utility in dye manufacturing, polymer synthesis, and organic chemistry. Its combination of nucleophilic amino groups, sterically active methyl substituents, and stable aromatic framework makes it an important building block for functional materials and bioactive compounds. References 2024. Polyimide-derived carbon molecule sieve membranes for gas separations. Nature Chemical Engineering. DOI: 10.1038/s44286-023-00021-1 2023. Precise molecular sieving of ethylene from ethane using triptycene-derived submicroporous carbon membranes. Nature Materials. DOI: 10.1038/s41563-023-01629-7 2022. Advanced carbon molecular sieve membranes derived from molecularly engineered cross-linkable copolyimide for gas separations. Nature Materials. DOI: 10.1038/s41563-022-01426-8 |
| Market Analysis Reports |