Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shijiazhuang Lida Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lid-ff.cn | |||
 | +86 (311) 8573-7996 | |||
 | +86 (311) 8573-9175 | |||
 | service@lid-ff.cn | |||
| Chemical manufacturer since 1993 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Dishman Pharmaceuticals & Chemicals Limited | USA | |||
|---|---|---|---|---|
 | www.dishmangroup.com | |||
 | +1 (732) 560-4300 | |||
 | +1 (732) 560-4343 | |||
 | bhaveshoza@dishmangroup.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Jinan Yudong Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yudong.com.cn | |||
 | +86 (531) 8231-2885 +86 15990993651 | |||
 | +86 (531) 8231-2887 | |||
 | yudong@yudong.com.cn happyxujuan@hotmail.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Jinan Great Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemgreat.com | |||
 | +86 (531) 5866-8191 | |||
 | +86 (531) 8632-7119 | |||
 | sale001@chemgreat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Weifang Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qianjinchemical.com | |||
 | +86 (536) 517-0190 | |||
 | +86 (536) 889-7109 | |||
 | 1297429207@qq.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Wuhan Lwax Pharma Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | whlawx.en.made-in-china.com | |||
 | +86 1313 567 7601 | |||
 | hanna@whlwax.com | |||
| Chemical distributor since 2021 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Wuhan Mulei New Materials Technology Co, Ltd. | China | |||
|---|---|---|---|---|
 | www.whmulei.com | |||
 | +86 (198) 3195-3957 | |||
 | jane@whmulei.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Honyan Pharmaceutical (wuhan) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hoyanmedicine.com | |||
 | +86 15832900195 | |||
 | +86 (27) 8233-9698 | |||
 | grace@whhoyan.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 008615832900195 | |||
 | WhatsApp:008615832900195 | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Wuhan Ruichi Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | whrchem.com | |||
 | +86 13545065237 | |||
 | admin@whrchem.com | |||
 | WhatsApp:+8613545065237 | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Zhejiang Rongchuang International Trade Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.peptideaas.com | |||
 | +86 15924248838 | |||
 | mihui41770@gmail.com | |||
| Chemical distributor since 2026 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Tongling Yangguang Synthetic Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sk-chem.com | |||
 | +86 (562) 582-7012 | |||
 | +86 (562) 582-7010 | |||
 | sales@sk-chem.com | |||
| Chemical manufacturer | ||||
| Chem Service, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemservice.com | |||
 | +1 (610) 692-3026 | |||
 | +1 (610) 692-8729 | |||
 | info@chemservice.com | |||
| Chemical manufacturer since 1962 | ||||
| Trigona oHG | Germany | |||
|---|---|---|---|---|
 | www.trigona.de | |||
 | +49 (611) 962-5283 | |||
 | +49 (611) 962-9032 | |||
 | info@trigona.de | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| DeLong Chemicals America | USA | |||
|---|---|---|---|---|
 | www.delongchemicals.com | |||
 | +1 (203) 271-9017 | |||
 | sales@delongchemicals.com | |||
| Chemical manufacturer | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Classification | Flavors and spices >> Synthetic spice >> Lactone and oxygen-containing heterocyclic compound >> Thiazole, thiophene and pyridine |
|---|---|
| Name | Tetrahydro pyrrole |
| Synonyms | Pyrrolidine |
| Molecular Structure | 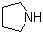 |
| Molecular Formula | C4H9N |
| Molecular Weight | 71.12 |
| CAS Registry Number | 123-75-1 |
| EC Number | 204-648-7 |
| FEMA | 3523 |
| SMILES | C1CCNC1 |
| Density | 0.8$+/-$0.1 g/cm3 Calc.*, 0.856 g/mL (Expl.) |
|---|---|
| Melting point | -63 $degree$C (Expl.) |
| Boiling point | 89.5$+/-$8.0 $degree$C 760 mmHg (Calc.)*, 87 - 88 $degree$C (Expl.) |
| Flash point | 2.8 $degree$C (Calc.)*, 2 $degree$C (Expl.) |
| Solubility | Soluble in alcohol, ether, chloroform. Miscible with water (Expl.) |
| Index of refraction | 1.428 (Calc.)*, 1.443 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS02;GHS05;GHS07 DangerGHS02; Details GHS02;GHS05;GHS07 DangerGHS02; Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H302-H314-H318-H332 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P261-P264-P264+P265-P270-P271-P280-P301+P317-P301+P330+P331-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P321-P330-P363-P370+P378-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1922 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Tetrahydropyrrole, more commonly known as pyrrolidine, is a saturated five-membered nitrogen-containing heterocycle with the molecular formula C4H9N. It can be viewed as the fully hydrogenated derivative of pyrrole, in which the aromaticity of the ring has been lost and replaced by a flexible, saturated structure. This compound has been known since the nineteenth century and has become an important building block in organic chemistry, pharmaceuticals, and industrial applications. The molecular structure of tetrahydropyrrole consists of a five-membered ring containing four carbon atoms and one nitrogen atom. Unlike pyrrole, which is aromatic, pyrrolidine is non-aromatic and adopts a puckered conformation that reduces angle strain. The nitrogen atom carries a lone pair of electrons, making the compound a secondary amine with significant basicity and nucleophilicity. This property allows it to participate readily in a wide range of chemical reactions, including alkylation, acylation, and condensation processes. Tetrahydropyrrole is commonly prepared by the hydrogenation of pyrrole in the presence of a suitable catalyst, a method that has been well established in the literature. It can also be synthesized from 1,4-diaminobutane through cyclization reactions or by reduction of suitable precursors. These methods provide efficient routes to the compound on both laboratory and industrial scales, yielding a stable liquid that is miscible with water and many organic solvents. One of the most important applications of tetrahydropyrrole is in organic synthesis, where it serves as a base and nucleophilic catalyst. It is widely used in the formation of enamines when reacted with carbonyl compounds. Enamines are key intermediates in many carbon–carbon bond-forming reactions, such as the Stork enamine reaction, allowing selective functionalization of aldehydes and ketones. This role has made pyrrolidine an essential reagent in synthetic organic chemistry. In medicinal chemistry, tetrahydropyrrole is a common structural motif found in numerous bioactive compounds and pharmaceuticals. The pyrrolidine ring is present in natural products and synthetic drugs, where it contributes to molecular rigidity, basicity, and the ability to interact with biological targets through hydrogen bonding and ionic interactions. As a result, pyrrolidine derivatives are used in the design of enzyme inhibitors, receptor ligands, and other therapeutic agents. Tetrahydropyrrole is also used in the production of agrochemicals, rubber chemicals, and corrosion inhibitors. Its reactivity and ability to form stable derivatives make it a versatile intermediate in industrial chemistry. Additionally, it can serve as a ligand in coordination chemistry, binding to metal centers through the nitrogen atom and influencing catalytic activity or stability of metal complexes. Overall, tetrahydropyrrole is a fundamental nitrogen-containing heterocycle with broad applications in synthesis, industry, and pharmaceutical development. Its combination of basicity, nucleophilicity, and structural flexibility makes it a valuable reagent and building block in modern chemistry. References 2025. Plant proteins for human and environmental health: knowledge, barriers, and levers for their development, a case study in France. A review. Agronomy for Sustainable Development. DOI: 10.1007/s13593-025-01034-1 2025. Synthesis of Fully Substituted 1,2,3-Triazoles via Organocatalytic [3+2] Cycloaddition: Incorporation of Pyrazole and Imidazole Scaffolds as Potent EGFR-Targeted Anticancer Agents. Russian Journal of Bioorganic Chemistry. DOI: 10.1134/s1068162024606633 2025. Synthesis of highly strained para-cyclophanes via ring-expansion [5,5]-sigmatropic rearrangement reaction. Nature Chemistry. DOI: 10.1038/s41557-025-01878-w |
| Market Analysis Reports |