Online Database of Chemicals from Around the World
| Nanjing Elegant Nutraceuticals Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (25) 8324-1163 | |||
 |
sales@elegantnutri.com machine@elegantnutri.com | |||
 |
QQ chat | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2006 | ||||
| Choriway International Trade Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (510) 8260-6408 | |||
 |
sales@choriway.com | |||
| Chemical distributor | ||||
| chemBlink standard supplier since 2007 | ||||
| Nantong Chem-land Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (513) 8551-2619 | |||
 |
sales@chem-land.com | |||
| Chemical distributor | ||||
| chemBlink standard supplier since 2007 | ||||
| Asia Talent Chemical Limited | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (755) 8665-5561 | |||
 |
jeffreyxu@atchem.net sinochemxu@hotmail.com asiatalentchem@gmail.com | |||
 |
Skype Chat | |||
 |
QQ chat | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| Zhangjiagang Sunrise Fine Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (574) 8797-0832 | |||
 |
info@sfcc.com.cn | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink standard supplier since 2008 | ||||
| Simagchem Corporation | China | Inquire | ||
|---|---|---|---|---|
 |
+86 13806087780 | |||
 |
sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink standard supplier since 2008 | ||||
| Changzhou Tianhua Pharmaceutical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (519) 8289-1186 | |||
 |
tianhua@tianhuachem.com ctianhua@hotmail.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink standard supplier since 2008 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 |
sales@pharm-intermediates.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink standard supplier since 2009 | ||||
| Classification | API >> Circulatory system medication >> Prevention and treatment of angina pectoris |
|---|---|
| Name | Minoxidil |
| Synonyms | 2,4-Diamino-6-piperidino-pyrimidine-3-oxide; 6-(1-Piperidinyl)-2,4-pyrimidinediamine-3-oxide; Loniten; Regain; Rogaine |
| Molecular Structure | 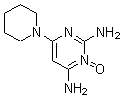 |
| Molecular Formula | C9H15N5O |
| Molecular Weight | 209.25 |
| CAS Registry Number | 38304-91-5 (16317-69-4) |
| EC Number | 253-874-2 |
| SMILES | C1CCN(CC1)C2=NC(=N)N(C(=C2)N)O |
| Solubility | 2.295e+004 mg/L (25 ºC water) |
|---|---|
| Density | 1.5±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.724, Calc.* |
| Melting point | 177.26 ºC |
| Boiling Point | 351.7±45.0 ºC (760 mmHg), Calc.*, 424.35 ºC |
| Flash Point | 166.5±28.7 ºC, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |
 GHS07 Warning Details
GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hazard Statements | H302-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||
| Precautionary Statements | P261-P280-P305+P351+P338 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Originally developed in the 1950s as an oral medication for the treatment of ulcers, researchers serendipitously discovered Minoxidil's unexpected side effect of promoting hair growth. This unexpected finding led to extensive research into the compound's mechanism of action and its potential application in addressing hair loss disorders. In the late 1970s, Minoxidil was repurposed as a topical solution for the treatment of androgenetic alopecia, commonly known as male and female pattern baldness. Minoxidil is widely used as a topical treatment for androgenetic alopecia in both men and women. Applied directly to the scalp, it stimulates hair follicles, prolongs the growth phase of the hair cycle, and increases hair density and thickness, leading to visible regrowth of hair in affected areas. Minoxidil has shown efficacy in the treatment of alopecia areata, an autoimmune condition characterized by patchy hair loss. Although its precise mechanism in alopecia areata is not fully understood, Minoxidil's ability to promote hair growth makes it a valuable therapeutic option for individuals with this condition. Following childbirth, many women experience temporary hair shedding known as postpartum telogen effluvium. Minoxidil can help accelerate the regrowth of hair and restore hair density in women experiencing postpartum hair loss. Minoxidil has gained popularity as an off-label treatment for stimulating beard growth in individuals with sparse or patchy facial hair. Minoxidil may also be used to treat other scalp conditions such as telogen effluvium, scalp psoriasis, and seborrheic dermatitis, where hair loss or thinning is a prominent feature. Its ability to promote hair regrowth and improve scalp health makes it a versatile therapeutic option for various scalp disorders. References 2024. The History of Hair Loss Treatments. Updates in Clinical Dermatology. DOI: 10.1007/978-3-031-74314-6_1 1984. Determination of Minoxidil in Bulk Drug and Pharmaceutical Formulations by Ion-Pairing High-Performance Liquid Chromatography. Journal of Pharmaceutical Sciences, 73(9). DOI: 10.1002/jps.2600730926 1987. Sulfation of minoxidil by human platelet sulfotransferase. Clinica chimica acta; international journal of clinical chemistry, 171(2-3). DOI: 10.1016/0009-8981(87)90322-6 |
| Market Analysis Reports |
| List of Reports Available for Minoxidil |