Online Database of Chemicals from Around the World
| Yangzhou Lianhuan Pharmaceutical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (514) 8782-9985 | |||
 |
scb@lhpharma.com api@lhpharma.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 8558-6718 +86 13336195806 | |||
 |
capotchem@gmail.com sales@capotchem.com | |||
 |
QQ chat | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2006 | ||||
| Zhejiang Esun Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (571) 5628-6531 | |||
 |
esunchem@gmail.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| Changzhou Share Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (519) 8261-9998 | |||
 |
wu@sharechem.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| Jinan Chenghui-Shuangda Chemical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (531) 5889-7051 +86 15053146086 | |||
 |
jnchsd@qq.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink standard supplier since 2007 | ||||
| Shanghai Longschem Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (21) 3635-6756 | |||
 |
charles-hu@longschem.com | |||
| Chemical manufacturer | ||||
| chemBlink standard supplier since 2007 | ||||
| Simagchem Corporation | China | Inquire | ||
|---|---|---|---|---|
 |
+86 13806087780 | |||
 |
sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink standard supplier since 2008 | ||||
| Changzhou Tianhua Pharmaceutical Co., Ltd. | China | Inquire | ||
|---|---|---|---|---|
 |
+86 (519) 8289-1186 | |||
 |
tianhua@tianhuachem.com ctianhua@hotmail.com | |||
 |
QQ chat | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink standard supplier since 2008 | ||||
| Classification | API >> Digestive system medication |
|---|---|
| Name | Cetilistat |
| Synonyms | 2-(Hexadecyloxy)-6-methyl-4H-3,1-benzoxazin-4-one |
| Molecular Structure | 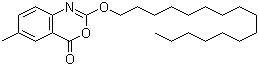 |
| Molecular Formula | C25H39NO3 |
| Molecular Weight | 401.58 |
| CAS Registry Number | 282526-98-1 |
| SMILES | CCCCCCCCCCCCCCCCOC1=NC2=C(C=C(C=C2)C)C(=O)O1 |
| Density | 1.0±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 509.7±43.0 ºC 760 mmHg (Calc.)* |
| Flash point | 158.9±22.6 ºC (Calc.)* |
| Index of refraction | 1.521 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |
 GHS07 Warning Details
GHS07 Warning Details |
|---|---|
| Hazard Statements | H302-H315-H319-H335 Details |
| Precautionary Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Cetilistat is a synthetic benzoxazinone derivative developed as a gastrointestinal lipase inhibitor for the treatment of obesity and associated metabolic disorders. It functions by reducing dietary fat absorption in the intestine, promoting weight loss in patients with obesity. Cetilistat is chemically distinct from orlistat, the first approved drug of this class, but it shares a similar mechanism of action through the inhibition of pancreatic lipase. The molecular structure of cetilistat includes a benzoxazinone core substituted with lipophilic side chains that contribute to its local activity in the gastrointestinal tract. The compound acts by covalently binding to the active serine site of pancreatic and gastric lipases, enzymes responsible for breaking down dietary triglycerides into absorbable free fatty acids and monoglycerides. By inactivating these enzymes, cetilistat prevents the hydrolysis and subsequent absorption of fats, resulting in increased excretion of unabsorbed triglycerides in feces. Cetilistat was originally developed by Alizyme and later licensed to Takeda Pharmaceutical Company. It was designed to improve upon the gastrointestinal tolerability profile of orlistat, which, although effective, is often associated with adverse effects such as oily spotting, fecal urgency, and flatulence due to excessive fat excretion. Clinical trials demonstrated that cetilistat provided significant weight loss in obese patients and improved glycemic control in those with type 2 diabetes mellitus, with a lower incidence of intolerable gastrointestinal side effects. The drug has been approved for the treatment of obesity in Japan under the trade name Oblean. It is indicated for obese patients, particularly those with obesity-related conditions such as impaired glucose tolerance, type 2 diabetes, and hyperlipidemia. Clinical studies conducted in Asia and elsewhere have shown that cetilistat, when administered at therapeutic doses in combination with a hypocaloric diet, leads to statistically significant reductions in body weight, waist circumference, and serum triglycerides compared to placebo. In some trials, its efficacy was comparable to orlistat, with improved patient adherence due to better gastrointestinal tolerability. Cetilistat is typically administered orally in capsule or tablet form. It is poorly absorbed into systemic circulation, which limits the potential for systemic side effects. Most of its pharmacological activity occurs locally in the lumen of the gastrointestinal tract. The unabsorbed portion is excreted in the feces along with unabsorbed dietary fats. The low systemic exposure also reduces the risk of interactions with other systemic medications. Common side effects of cetilistat are usually related to the gastrointestinal system and include steatorrhea, increased bowel movement, abdominal discomfort, and flatulence. These effects are generally mild to moderate and often diminish with continued use or dietary adjustment to reduce fat intake. As with all lipase inhibitors, the use of cetilistat may impair the absorption of fat-soluble vitamins (A, D, E, and K), and supplementation may be recommended during long-term therapy. Preclinical studies of cetilistat also investigated its potential role in improving insulin sensitivity and reducing inflammatory markers associated with obesity and metabolic syndrome. Although not approved for these indications, the compound’s primary benefit in weight reduction contributes indirectly to improvement in these conditions. Cetilistat is stable under standard storage conditions and is supplied in solid dosage forms for oral administration. Its chemical stability and limited absorption profile contribute to a favorable safety margin. The drug is not approved in all countries, and its availability is limited to specific markets where regulatory approval has been granted based on regional clinical data. In conclusion, cetilistat is a gastrointestinal lipase inhibitor used for the treatment of obesity. Its clinical efficacy in promoting weight loss and improving metabolic parameters, along with a better gastrointestinal tolerability profile compared to earlier agents, make it a significant option in pharmacologic obesity management, particularly in patients with comorbid metabolic disorders. References 2020. Discovery of the FDA-approved drugs bexarotene, cetilistat, diiodohydroxyquinoline, and abiraterone as potential COVID-19 treatments with a robust two-tier screening system. Pharmacological Research, 159. DOI: 10.1016/j.phrs.2020.104960 2019. Pharmacotherapy of Obesity: Limits and Perspectives. American journal of cardiovascular drugs : drugs, devices, and other interventions, 19(2). DOI: 10.1007/s40256-019-00328-6 2018. Future Pharmacotherapy for Obesity: New Anti-obesity Drugs on the Horizon. Current Obesity Reports, 7(2). DOI: 10.1007/s13679-018-0300-4 |
| Market Analysis Reports |
| List of Reports Available for Cetilistat |