Online Database of Chemicals from Around the World
| Dalian Richfortune Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.richfortunechem.com | |||
 | +86 (411) 8482-0922 8482-1539 | |||
 | +86 (411) 8482-1380 | |||
 | sales@richfortunechem.com lrxwl@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shandong Holon Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.holonchem.com | |||
 | +86 (533) 211-1311 | |||
 | +86 (533) 211-1311 | |||
 | admin@holonchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Wuhan Air Gas Electronic Materials Enterprise Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.whagem.com | |||
 | +86 18164072249 | |||
 | s5@whagem.com | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Advance Research Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.fluoridearc.com | |||
 | +1 (918) 266-6789 | |||
 | +1 (918) 266-6796 | |||
 | sales@fluoridearc.com | |||
| Chemical manufacturer | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Hydride, nitride, azide |
|---|---|
| Name | Nitrogen trifluoride |
| Synonyms | Trifluoramine |
| Molecular Structure | 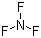 |
| Molecular Formula | F3N |
| Molecular Weight | 71.00 |
| CAS Registry Number | 7783-54-2 |
| EC Number | 232-007-1 |
| SMILES | N(F)(F)F |
| Density | 1.4$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | -206.7 $degree$C (Expl.) |
| Boiling point | -128.8$+/-$9.0 $degree$C 760 mmHg (Calc.)*, -128.9 $degree$C (Expl.) |
| Solubility | Slightly soluble (Expl.) |
| Index of refraction | 1.187 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |     GHS03;GHS04;GHS07;GHS08 Danger Details GHS03;GHS04;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H270-H280-H332-H373 Details | ||||||||||||||||||||||||
| Safety Statements | P220-P244-P260-P261-P271-P304+P340-P317-P319-P370+P376-P403-P410+P403-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Transport Information | UN 2451 | ||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Nitrogen trifluoride is a colorless, odorless, and nonflammable gas with the chemical formula NF3. It consists of a central nitrogen atom covalently bonded to three fluorine atoms in a trigonal pyramidal geometry. Nitrogen trifluoride was first prepared in the early 20th century and has gained industrial significance due to its chemical stability and strong oxidizing fluorine atoms, despite the inertness of the nitrogen center under normal conditions. The molecular structure of NF3 features sp3 hybridization at nitrogen, with a lone pair of electrons occupying one of the four tetrahedral positions, resulting in a trigonal pyramidal shape. The N–F bonds are highly polar due to the large electronegativity difference between nitrogen and fluorine, but the overall molecule is relatively stable and does not easily react with most substances under ambient conditions. It is thermally stable and only decomposes at high temperatures or under plasma conditions, releasing reactive fluorine radicals. Nitrogen trifluoride is widely used in the electronics industry as a cleaning and etching gas for semiconductor manufacturing. In plasma etching processes, NF3 is decomposed under radiofrequency or microwave energy to generate fluorine radicals, which selectively remove silicon, silicon dioxide, and silicon nitride from wafer surfaces without attacking metal layers. Its ability to produce reactive fluorine in a controlled manner makes it an essential chemical for producing integrated circuits, thin-film devices, and flat-panel displays. In addition to semiconductor applications, NF3 is used in the preparation of fluorine-containing compounds and as a fluorinating agent in chemical synthesis. Its inertness under standard conditions allows for safe storage and transport, while its decomposition under energy input provides a source of reactive species for high-precision processes. Environmental considerations are important for nitrogen trifluoride because it is a potent greenhouse gas with a very long atmospheric lifetime. Even in small concentrations, NF3 contributes significantly to radiative forcing, and efforts are made in industrial settings to capture and destroy emissions. Advanced abatement systems, such as plasma destruction and catalytic decomposition, are employed to minimize environmental impact during manufacturing. Overall, nitrogen trifluoride is a chemically stable, fluorine-containing gas with specialized applications in semiconductor manufacturing, chemical synthesis, and material processing. Its combination of inertness under ambient conditions and controlled reactivity under plasma or thermal activation makes it a valuable industrial chemical, while its environmental impact requires careful management. References 2025. Considering sectoral warming and cooling emissions and their lifetimes can improve climate change mitigation policies. npj Climate and Atmospheric Science. DOI: 10.1038/s41612-025-01131-8 2025. Green trading mechanisms, carbon taxation, and technology integration: achieving the carbon neutrality ambition in OECD countries. Clean Technologies and Environmental Policy. DOI: 10.1007/s10098-025-03185-x 2025. Varied drivers for reducing CO2 emissions in countries with legislated carbon neutrality targets: a Kaya identity clustering analysis. Environment, Development and Sustainability. DOI: 10.1007/s10668-025-06223-2 |
| Market Analysis Reports |