Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Fluoropharm Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoropharm.com | |||
 | +86 (571) 8558-6753 +86 13336034509 | |||
 | +86 (571) 8558-6753 | |||
 | sales@fluoropharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Wuhan Air Gas Electronic Materials Enterprise Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.whagem.com | |||
 | +86 18164072249 | |||
 | s5@whagem.com | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Organic raw materials >> Organic fluorine compound >> Fluoroethane series |
|---|---|
| Name | Hexafluoroethane |
| Synonyms | 1,1,1,2,2,2-Hexafluoroethane; Ethyl hexafluoride; F 116; F 116 (fluorocarbon); FC 116; FC 1160; Freon 116; HFC 116; Hexafluoroethane; PFC 116; Perfluorocarbon 116; Perfluoroethane; R 116 |
| Molecular Structure | 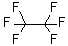 |
| Molecular Formula | C2F6 |
| Molecular Weight | 138.01 |
| CAS Registry Number | 76-16-4 |
| EC Number | 200-939-8 |
| SMILES | C(C(F)(F)F)(F)(F)F |
| Density | 1.5$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | -100 $degree$C (Expl.) |
| Boiling point | -82.3$+/-$8.0 $degree$C 760 mmHg (Calc.)*, -78 $degree$C (Expl.) |
| Flash point | -60.9$+/-$10.4 $degree$C (Calc.)* |
| Index of refraction | 1.203 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS04 Warning Details GHS04 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H280 Details | ||||||||||||
| Safety Statements | P410+P403P410+P403 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| Transport Information | UN 2193 2.2 | ||||||||||||
| SDS | Available | ||||||||||||
|
Hexafluoroethane is a colorless, odorless, and non-flammable gas with the chemical formula C2F6. It belongs to the class of perfluorocarbons, where all hydrogen atoms in ethane are replaced by fluorine atoms. This full fluorination imparts exceptional chemical stability, thermal resistance, and inertness, making hexafluoroethane resistant to oxidation, acids, bases, and most reducing agents. It was first characterized in the mid-20th century as part of the study of fluorinated hydrocarbons and has since been used in a variety of industrial and technological applications. The molecular structure of hexafluoroethane consists of two carbon atoms single-bonded to each other, with each carbon fully substituted by three fluorine atoms. The strong carbon–fluorine bonds give the molecule a high bond energy and contribute to its low chemical reactivity. Its tetrahedral geometry around each carbon atom results in a symmetric, non-polar molecule. Hexafluoroethane is gaseous at standard temperature and pressure, with a boiling point of –78.2 °C and a melting point of –100 °C, reflecting weak intermolecular van der Waals interactions. Hexafluoroethane is widely used as a refrigerant in specialized cooling systems, often labeled as R-116, due to its stability and inertness. Its non-flammable nature and low toxicity make it a preferred option in industrial refrigeration, though environmental concerns have limited its use because it is a potent greenhouse gas with a long atmospheric lifetime. It is also employed as a pressurizing gas in electronics manufacturing, where inert atmospheres are needed to prevent oxidation or contamination of sensitive materials. In semiconductor manufacturing, hexafluoroethane is used as an etchant gas in plasma etching processes. Its chemical inertness allows it to contribute fluorine radicals under plasma conditions, which selectively etch silicon-containing materials while maintaining precision and minimizing unwanted reactions. This application is critical in the fabrication of integrated circuits, microelectromechanical systems, and other microfabricated devices. Due to its chemical stability and low reactivity, hexafluoroethane has also been used in scientific research, such as in gas chromatography as a carrier gas or in calibration of instruments requiring non-reactive reference gases. Its strong greenhouse potential and global warming impact have led to research into alternatives and emission controls in industrial settings. Overall, hexafluoroethane is a chemically inert, thermally stable perfluorocarbon with applications in refrigeration, electronics manufacturing, and scientific research. Its full fluorination provides high stability and non-reactivity, while environmental considerations limit its broader use due to its potency as a greenhouse gas. References 2025. Bird’s-eye view of per- and polyfluoroalkyl substances pollution research in the African hydrosphere. npj Clean Water. DOI: 10.1038/s41545-025-00495-x 2025. Carbon footprint of Pars plana vitrectomies in rhegmatogenous retinal detachment in Germany between 2013 and 2023 - A cohort study of fluorinated gas emissions. Graefe's archive for clinical and experimental ophthalmology = Albrecht von Graefes Archiv fur klinische und experimentelle Ophthalmologie. DOI: 10.1007/s00417-025-06905-7 2025. Predicting post-surgery change in visual acuity after successful repair of macula-off retinal detachments: findings from a large prospective UK cohort study. Eye (London, England). DOI: 10.1038/s41433-025-03752-y |
| Market Analysis Reports |