Online Database of Chemicals from Around the World
| Zhangjiagang For Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.forchemical.com | |||
 | +86 (512) 5679-6585 5897-6331 | |||
 | +86 (512) 5897-6331 | |||
 | info@forchemical.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhejiang Rongchuang International Trade Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.peptideaas.com | |||
 | +86 15924248838 | |||
 | mihui41770@gmail.com | |||
| Chemical distributor since 2026 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Aronis | Russia | |||
|---|---|---|---|---|
 | www.aronis.ru | |||
 | +7 (926) 578-0336 | |||
 | +7 (495) 426-5336 | |||
 | rakishev@aronis.ru | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives |
|---|---|
| Name | 3-Amino-4-phenylbutyric acid hydrochloride |
| Synonyms | 4-amino-3-phenylbutanoic acid;hydrochloride |
| Molecular Structure | 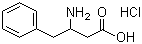 |
| Molecular Formula | C10H14ClNO2 |
| Molecular Weight | 215.68 |
| CAS Registry Number | 3060-41-1 |
| EC Number | 640-227-8 |
| SMILES | C1=CC=C(C=C1)C(CC(=O)O)CN.Cl |
| Melting point | 194-201 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
3-Amino-4-phenylbutyric acid hydrochloride is an amino acid derivative in which a primary amino group is positioned at the 3-carbon of a butyric acid backbone and a phenyl group is attached at the 4-carbon. The hydrochloride form enhances its solubility and stability, making it suitable for use in pharmaceutical and biochemical applications. The compound combines the reactivity of the amino group with the hydrophobic characteristics of the phenyl moiety, enabling interactions in both chemical and biological systems. The molecular structure consists of a linear four-carbon chain bearing a carboxylic acid at one end, an amino group at the third carbon, and a phenyl substituent at the fourth carbon. The hydrochloride salt ensures that the amino group is protonated, increasing water solubility and facilitating handling in aqueous media. This structural arrangement also allows the compound to participate in peptide bond formation and other condensation reactions, which is important for its role as a chemical intermediate. Synthesis of 3-amino-4-phenylbutyric acid hydrochloride typically involves the functionalization of a suitable phenylbutyric acid precursor. Strategies include selective amination at the 3-position or reduction of corresponding nitro or oxo intermediates followed by conversion to the hydrochloride salt. Careful control of reaction conditions is necessary to maintain regioselectivity and preserve the stereochemistry, particularly if the compound is used in biological applications. The compound has applications in medicinal chemistry, where it can serve as a building block for bioactive molecules. Its amino and carboxyl groups allow for incorporation into peptide analogs or modified amino acids, while the phenyl group contributes to hydrophobic interactions that can influence binding to enzymes or receptors. Such properties have led to its investigation as an intermediate in the synthesis of neurotransmitter analogs and other pharmacologically relevant compounds. In addition to pharmaceutical applications, 3-amino-4-phenylbutyric acid hydrochloride is used in organic synthesis as a versatile intermediate. Its functional groups permit derivatization, including acylation, alkylation, or coupling reactions, allowing chemists to prepare more complex molecules with tailored chemical or physical properties. The hydrochloride salt form ensures stability and ease of handling, which is particularly useful in multi-step synthetic sequences. Overall, 3-amino-4-phenylbutyric acid hydrochloride is a multifunctional amino acid derivative with significant utility in pharmaceutical and synthetic chemistry. Its combination of an amino group, carboxylic acid, and phenyl substituent provides chemical versatility, enabling its use as an intermediate in drug development, peptide chemistry, and other specialized chemical syntheses. References 2024. Phenibut: A drug with one too many “buts”. Basic & Clinical Pharmacology & Toxicology. DOI: 10.1111/bcpt.14075 2024. Phenibut: Review and Pharmacologic Approaches to Treating Withdrawal. The Journal of Clinical Pharmacology. DOI: 10.1002/jcph.2414 2024. Phenibutan—an Illegal Food Supplement With Psychotropic Effects and Health Risks. Deutsches Ärzteblatt international. DOI: 10.3238/arztebl.m2024.0003 |
| Market Analysis Reports |