Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wuhan Air Gas Electronic Materials Enterprise Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.whagem.com | |||
 | +86 18164072249 | |||
 | s5@whagem.com | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | API >> Nervous system medication >> Central stimulant |
|---|---|
| Name | Carbon dioxide |
| Synonyms | Baros Effervescent Granules; Carbon oxide (CO2); Carbon-12 dioxide; Carbon-12C dioxide-16O2; Carbonic acid anhydride; Carbonic acid gas; Carbonic anhydride; Dry ice; EN 439C1; Khladon 744; N 45 (gas); R 744 |
| Molecular Structure | 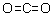 |
| Molecular Formula | CO2 |
| Molecular Weight | 44.01 |
| CAS Registry Number | 124-38-9 |
| EC Number | 204-696-9 |
| SMILES | C(=O)=O |
| Density | 1.799 g/cm3 (25 °C)* |
|---|---|
| Melting point | 60-61 °C (ethanol )** |
| Boiling point | -78 °C (800 Torr)*** |
| Refractive index | 1.1839 (40.00 °C)***** |
| Flash point | -131.1±13.9 °C, Calc.**** |
| * | Molina, Alejandro |
| ** | Koshechko, V. G. |
| *** | Lebedev, A. V. |
| **** | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2016 ACD/Labs) |
| ***** | Sun, Yongda |
| Hazard Symbols |  GHS04 Warning Details GHS04 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H280-H281 Details | ||||||||||||||||||||||||||||
| Safety Statements | P282-P336+P317-P403-P410+P403 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 1013 - class 2 - Carbon dioxide | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Carbon dioxide is a simple inorganic compound composed of one carbon atom covalently bonded to two oxygen atoms, with the molecular formula CO2. It is a colorless, odorless gas under standard conditions and occurs naturally in Earth's atmosphere as a trace gas. Carbon dioxide has been known since the seventeenth century, when it was first described as “fixed air” by early chemists studying gases produced by fermentation and combustion processes. It is a linear molecule with the carbon atom in the center, forming double bonds with each oxygen atom, and exhibits resonance structures that distribute electron density across the molecule. Carbon dioxide plays a critical role in biological and environmental processes. It is a key component of the carbon cycle, produced by respiration in animals, fermentation in microorganisms, and the combustion of organic matter. Plants, algae, and certain bacteria utilize carbon dioxide in photosynthesis, converting it into organic molecules and oxygen. This natural exchange maintains the balance of atmospheric gases and supports life on Earth. Industrial production of carbon dioxide occurs primarily as a byproduct of combustion of fossil fuels, fermentation processes, and chemical reactions such as the reaction of acids with carbonates. Commercially, it is collected, purified, and compressed for various applications. Carbon dioxide is widely used in the food and beverage industry as a carbonation agent for soft drinks and sparkling water. Its non-toxic nature at controlled concentrations makes it suitable for these purposes, and it also acts as a preservative and cooling agent in its solid form as dry ice. In chemical synthesis, carbon dioxide is an important reagent and feedstock. It can react with nucleophiles to form carboxylates, carbonates, carbamates, and ureas. Its incorporation into organic molecules provides a route to synthesize valuable compounds such as salicylic acid, cyclic carbonates, and certain pharmaceuticals. Carbon dioxide is also employed as a solvent in supercritical fluid extraction, where supercritical CO2 is used for decaffeination of coffee, extraction of essential oils, and other processes requiring selective solubility and mild conditions. Carbon dioxide has significant environmental relevance. It is a greenhouse gas, trapping heat in the atmosphere and contributing to global warming. Human activities, particularly the burning of fossil fuels and deforestation, have increased atmospheric concentrations of CO2, intensifying the greenhouse effect and leading to climate change. Monitoring and controlling CO2 emissions is a major focus of environmental science and policy, and carbon capture and storage technologies are being developed to mitigate its impact. In addition to industrial and environmental roles, carbon dioxide is used in fire suppression systems, as a refrigerant, and as a propellant in aerosol applications. Its physical properties, such as sublimation to dry ice at atmospheric pressure, allow for versatile applications in cooling, cleaning, and preservation. Overall, carbon dioxide is a fundamental inorganic molecule with critical importance in biology, industry, and environmental science. Its dual role as both a necessary component of life processes and a contributor to climate change underscores its significance across multiple fields of study. References 2025. Remodeling aboveground tomato plant architecture via CRISPR/Cas9-mediated editing of a single Tiller Angle Control 1–like gene. aBIOTECH. DOI: 10.1007/s42994-025-00220-9 2025. The role of isoprenoids in the chemical interaction between plants and other organisms in their rhizosphere. aBIOTECH. DOI: 10.1007/s42994-025-00225-4 2025. Correction: Lacosamide Is a Novel Drug That Improves AGTPBP1 Knockout-Mediated Impairment of Neuronal and Dopaminergic Function. Molecular Neurobiology. DOI: 10.1007/s12035-025-05016-y |
| Market Analysis Reports |